- HOME
- New Product and Technology
- Cathepsin D/E Substrate MOCAc-Gly-Lys-Pro-Ile-Leu-Phe- Phe-Arg-Leu-Lys(Dnp)-D-Arg-NH2
Cathepsin D/E Substrate MOCAc-Gly-Lys-Pro-Ile-Leu-Phe- Phe-Arg-Leu-Lys(Dnp)-D-Arg-NH2
Intracellular proteases are focused for studying a control system of the cellular function. Many proteases, like cathepsins, calpains, caspases, and enzymes for ubiquitin-proteasome system, are controlling the cellular functions with interaction each other.
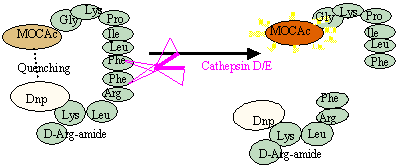
Cathepsin E and cathepsin D are two major intracellular aspartic proteinases implicated in the physiological and pathological degradation of intra- and extracellular proteins. Thus far, there are no useful synthetic substrates provide with a simple and fast method for measuring enzyme activities of large numbers of samples.
Recently, Yasuda et al. reported new fluorogenic substrate for the rapid and sensitive assay of cathepsin E and cathepsin D [J. Biochem. 125, 1137 (1999)]. This substrate is hydrolyzed equally well by cathepsins E and D (kcat/Km = 10.9 mM-1 · s-1 for cathepsin E and 15.6 mM-1 · s-1 for cathepsin D). This substrate represents a potentially valuable tool for mechanistic studies on cathepsins E and D.
| Code | Compound | Package |
| 3200-v | MOCAc-Gly-Lys-Pro-Ile-Leu-Phe-Phe-Arg-Leu-Lys(Dnp)-D-Arg-NH2 | 1 mg vial |

